Glaucoma
Glaucoma is recognizable by typical optic nerve damage.
In advanced glaucoma, there is considerable loss of visual field and even blindness before visual acuity is lost. Globally, glaucoma is the second most common cause of blindness after cataract. It is assumed that approximately 2.5% of the Swiss population over 40 years of age suffer from glaucoma.
-
CAUSES, RISK FACTORS
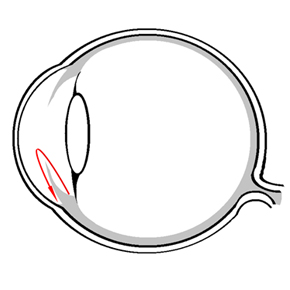
The cause of glaucoma is considered to be an imbalance between the intraocular pressure and blood flow to the optic nerve, which leads to nerve fiber damage. However, the exact cause has not been conclusively determined. The intraocular pressure results from the balance between production and outflow of the aqueous humor (intraocular fluid). The aqueous humor is produced in the ciliary body located behind the iris and flows through the pupil into the anterior chamber of the eye (Figure 1). There, the aqueous humor is drained via the trabecular meshwork in the chamber angle (structure between cornea and iris). The most common form of glaucoma results from a too high outflow resistance in the area of the trabecular meshwork, which leads to an intraocular pressure that is no longer tolerated by the optic nerve and is therefore too high. The increased intraocular pressure leads to a reduced blood flow (e.g. due to too low blood pressure or temporary vascular constriction) with the long-term consequence of damage to the optic nerve.
A distinction is made between open and closed-angle glaucoma.
In open angle glaucoma (by far the most common form in Central Europe) there is too much outflow resistance even with open outflow (chamber angle).
In closed angle glaucoma (much more common in people of Asian descent), there is a temporary or permanent narrowing of the chamber angle. This results in an increased outflow resistance and an increase in intraocular pressure. Acute glaucoma is a subtype of closed angle glaucoma. A strong and usually very painful increase in intraocular pressure usually occurs within a few hours. A glaucoma attack represents an emergency and is usually accompanied by severe eye and headaches. In addition to a reddened eye, general symptoms such as nausea, vomiting, abdominal pain and a general feeling of illness can also occur. Therefore, any suspicion of a glaucoma attack requires a quick ophthalmological examination and treatment in order to keep the risk of permanent damage to the eye low.General risk factors for glaucoma are:
- Increased intraocular pressure (IOP): The statistical normal range of intraocular pressure is between 10 and 21 mmHg (millimeters of mercury). Higher IOP means a higher risk of developing glaucoma, but there are rarely people with higher IOP who do not have long-term glaucoma damage. Conversely, there are a large number of patients (estimates suggest up to 50%) with typical permanent glaucomatous changes despite consistently presenting normal intraocular pressure values (so-called normal pressure glaucoma).
- According to studies, glaucoma in a close relative increases the risk of developing glaucoma two to four times.
- Pseudoexfoliation (also called capsular membrane): The so-called pseudoexfoliation describes the deposition of protein material in the anterior segment of the eye. The protein deposits in the chamber angle can lead to obstruction of the aqueous humor outflow. As a result, the eye pressure fluctuates more and more and can rise dramatically in the long term, which is why the risk of damage to the optic nerve and visual field is particularly high.
Further risk factors are:
- High or even very low blood pressure
- Age
- Myopia
- Low central corneal thickness
-
SYMPTOMS
Chronic open-angle glaucoma is typically a painless eye disease, which is often only detected when there has already been a marked loss of vision.
For this reason, a regular ophthalmological examination for early detection is recommended from the age of 40 or earlier in patients with a positive family history (relatives with glaucoma).Acute glaucoma occurs mainly in farsighted eyes and is usually accompanied by severe eye and headaches, so it is rarely missed.
-
DIAGNOSTICS
In order to make a diagnosis of glaucoma, the ophthalmologist uses various examination methods. These include taking the patient’s medical history, querying risk factors, measuring the intraocular pressure and assessing the anterior and posterior segment of the eye and the chamber angle. The examination is completed by further instrumental examinations such as visual field examination, measurement of corneal thickness and scanning laser ophthalmoscopy to measure the amount of nerve fibers.
Photo of a normal (top) and a glaucomatous optic nerve (bottom; vessels pushed to the side, central brightening due to nerve fiber damage), including the corresponding visual field examinations.
-
TREATMENT
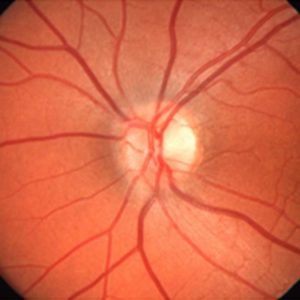 Photo of a normal (top) and a glaucomatous optic nerve (bottom; vessels pushed to the side, central brightening due to nerve fiber damage), including the corresponding visual field examinations.
Photo of a normal (top) and a glaucomatous optic nerve (bottom; vessels pushed to the side, central brightening due to nerve fiber damage), including the corresponding visual field examinations.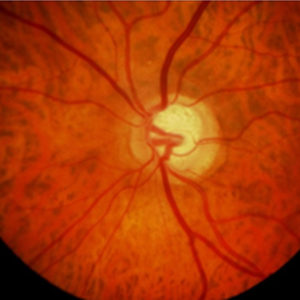
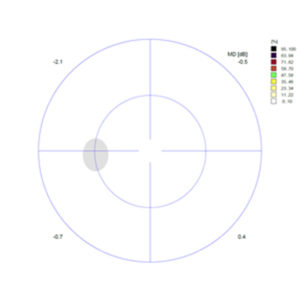
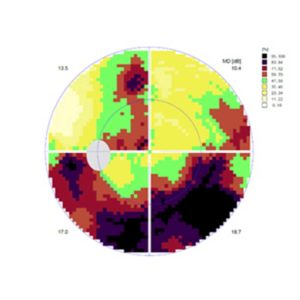
The color changes show how severe the loss of contrast vision is in the respective area of the field of vision; where it is black, the eye cannot recognize even bright light impulses.
The regular eye examinations serve to detect the disease at an early stage, if possible before structural optic nerve damage has occurred. The aim of treatment is to stop the disease from progressing. It is not possible to restore the damage that has already occurred (visual field failures). In order to stop the progression of the optic nerve damage, the main aim is to reduce the intraocular pressure. Over the course of time, each patient must individually strive for a level of intraocular pressure at which no further progression of the damage is apparent (target pressure). To achieve this, a drug therapy with one or more eye drops is usually applied at the beginning. If the eye drops do not respond adequately or are intolerant, laser therapy can often be performed first (e.g. selective laser trabeculoplasty, argon laser trabeculoplasty, YAG iridotomy, iridoplasty).
If no sufficient effect can be achieved by eye drop or laser therapy or if the patient has very advanced glaucoma, a surgical procedure is necessary. There are various procedures available:
- Laser-assisted procedures (e.g., cyclophotocoagulation, micropulse cyclophotocoagulation): here, either from the outside or from the inside (e.g. within the scope of a cataract operation), laser energy is used to damage the ciliary body that forms the aqueous humor to such an extent that it produces less intraocular fluid (aqueous humor).
- Minimally invasive procedures, e.g. from interno trabeculectomy, implantation of various implants (e.g. iStent, XEN-Stent): the aim is to improve the outflow of aqueous humor. These procedures can also be combined with cataract surgery.
- Trabeculectomy: The oldest operation against glaucoma and still the gold standard for many ophthalmologists. In this operation, an additional drainage path is created between the anterior chamber of the eye and the conjunctiva, which creates a so-called filtering bleb of the excess fluid produced when it escapes from the eye.
- PRESERFLO Microshunt: after opening the conjunctiva, this microtube is inserted into the anterior chamber in a minimally invasive way. The goal is to achieve an additional drainage path between the anterior chamber and the conjunctiva, as in the classic trabeculectomy.
- Implantation of valves (e.g. Ahmed valve): This type of implantation is usually performed in patients for whom several previous surgeries have been unsuccessful or in case of special forms of glaucoma (glaucoma as a result of vascularization or inflammation of the eye).
-
After your operation
Contact us if you experience pain or sudden redness. The same applies if your vision suddenly deteriorates or if you notice shadows or flashes.
Vision may be somewhat blurred for about two days. A slight scratchy feeling, tearing or a sensation of a foreign body in the operated eye is normal in the first two days. Double vision and increased glare is also possible.
During the day, no bandage is necessary. You can protect the eye as you wish with glasses or sunglasses.
The operated eye is treated for several weeks with Spersacarpine and TobraDex eye drops.
Dosage (unless otherwise prescribed)
Shake well before use, one drop (at least 10 minutes apart) into the operated eye at approximately the following times:Spersacarpine for 4 weeks
8 am12 am4 pm8 pmTobraDex according to prescription ophthalmologist
8 am12 am4 pm8 pmYour ophthalmologist will tell you at your follow-up examination when you can reduce TobraDex dosage and when you can drive again. Then continue to administer your eye drops to both eyes in the evening as usual.
Permitted are:
- Showering and hair washing, keeping the operated eye closed. No soap or shampoo should enter the operated eye.
- Light work, general activities of daily life, bending, light gymnastics, lifting of loads up to 10kg.
- Walking, in sunny weather you can protect your eyes with sunglasses.
- Television, reading and writing as long as you feel comfortable.
Prohibited:
- Rubbing or pressing your eyes or squinting hard.
- Lifting of loads over 10kg in the first week.
- Working in the garden or stable during the first two weeks.
- Weight training and swimming in the first two weeks.
- Driving, until the permission is given by your treating ophthalmologist.
Follow-up examinations take place as agreed. Usually the controls are one day after the operation, then after one week and after one month. These examinations can be carried out by the referring ophthalmologist.
Newly prescribed glasses usually only makes sense after 4-6 weeks after the operation. Temporary glasses can be used during this time.
In case of emergency (also on Saturday, Sunday and on holidays) you can reach an ophthalmologist at our eye clinic at the following number:
031 311 12 22

